Cuttlefish bone in the aquarium? Sure! In this article we explain you why to use it and how to use it.
Everyone has experienced at least once a too low value of hardness (KH) in his acquarium. For example, because an allophane has absorbed too much salt; or because we exaggerated with osmosis water; or, in some cases, cause our tap water is too soft for the fishes we decided to have. At that point the question comes by itself: «How to increase the hardness?».
If our tap water is hard enough we change the water; however this is not the ideal solution during the maturation phase. Or we could use commercial salts; however most of them are rich in Sodium and expensive.
Luckily there is a simple, natural and economical solution that will solve our problem: the cuttlefish bone.

What is the cuttlebone?
Let’s start from the beginning: what is this bone?
As the name suggests, it is the skeleton of the cuttlefish, a cephalopod, as we all know.
The bone of this animal is full of gas and helps to control the buoyancy of the fish in the water. For years people have collected and used these bones for various purposes; the most common use is as a food supplement and a ”toy” for birds.
Cause cuttlefish is a very popular animal along our coastline, the bone is easy to find walking on the shore after storms

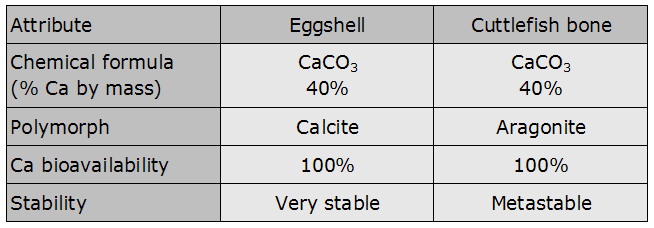
This shell is mostly made of calcium carbonate (CaCO3); Calcium represents 40% of its total weight. There are also other very important elements, such as Potassium, Iron and Zinc.
This carbonate is chemically defined as metastable; it is able to lose its stability after an energy supply.
Let’s talk about this in the next paragraph.
Eggshell vs cuttlefish bone
Chemistry teaches that eggshell and cuttlefish bones are both made of Calcium Carbonate. So, why should we prefer the second to the first one?
This table helps understanding the differences:
Both are mainly composed of Calcium Carbonate and in both cases calcium is available at 100%.
The difference is in that the egg is made of Calcite, while the bone is made of Aragonite.
But what does that mean?
Both are Calcium carbonates, but the substantial difference is their crystalline structure. The first is very stable, while the second is more «unstable», or better metastable. Due to this stability the eggshell melt less easier than the cuttlebone.
By trying to crush the eggshell, the powder will tend to stay in the water for longer and, moreover, the animals will eat it less.
To better understand the chemical difference between these two elements it may be useful to make a similarity with Graphite (for example of the pencils) and diamond: they are both made of Carbon, the same identical element. The difference is how Carbon binds itself: in Graphite it is arranged in planes flowing one over the other. In the diamond, however, this element forms an extremely strong, giant grid.
Where to buy the cuttlefish bone?
Why do I have to buy the bone when I can find it on the beach or in a fish shop?
The answer is simple: the bones collected on the beach or took in fish shops are covered with salt.
As we all know, in a freshwater aquarium salt should only be present in small quantities; that is why we will avoid the bones collected on the beach. The same argument applies to the ones in fish shops.
We can safely buy them at the supermarket, in the area dedicated to animals!

In the bird articles you will find this precious calcium source at the price of 2-3 euros for a couple of pieces.
Always check that the packaging is marked “washed and desalinated“.
As an old italian saying teaches:trusting is good, not trusting is better.
Now, the question is: why do we buy it?
To increase a too acid value of pH; to provide Calcium to the plants; to raise the hardness of the water; or to fix deficiencies in our gastropods.
Shells like those in the following pictures clearly indicate a lack of Calcium in the animal’s diet.


These deficiencies appear with a strange coloring on the snail shell, like stripes or white portions that feel rough when touched.
In the two images above, one can even see how shell growth has been difficult for the animal: apart from the whitish portions, there are also visible lines indicating the growth stages.
A perfect shell looks uniform in texture and color, not like the one in the picture.

The lack of Calcium, besides the Planorbella seen in the picture, affects any other gastropod present in our tanks, from Melanoids to Pomacea (Ampullaria).
In these last two kinds, such lack can be observed by looking at the initial part of the shell, the oldest part

The parts that are often lacking in animals affected by a lack of Calcium are those circled.
In this photo we can see how the Melanoides shows the effects of a lack of Calcium while the Pomacea is in perfect health.
How to use it?
Here it is the practical part of this article. Once we saw the deficiencies and we bought the bone … how do we use it? What do we do?
We can use its characteristic of giving calcium in different ways.
We can leave it in one piece, maybe in the filter compartment, as a “parachute” in case of a sudden drop of pH (as we have seen, sour water easily dissolves it and it goes back to neutral values).
Or we can crumble and pulverize it both for faster absorption by animals and for faster melting in water.
I recommend this second method because I have always observed a complete absorption overnight.
To do this, I use a bone and a long metal clamp.
Maybe you’re wondering why the long metal clamp … Actually there is no reason at all! I use the part where its two arms join just because it is very comfortable to rub and pulverize the bone!

I simply rub the wide part of the clamp against the porous part of the bone. Anything can be used for this purpose, from the non-cutting edge of a scissor to the handle of a spoon!
The result I get is this:

After grating part of it, about half or 1/3 of bone, I simply leave it lying on the surface of the water.


Once this is done, the job is over!
Basically the bone will absorb the water and fall to the bottom of the aquarium.
It’s pretty easy to see how snails take advantage of this powder right away, they eat it directly from the surface of the water.

After about one hour, it will look like the winter arrived in the tank with a beautiful snowfall!

Bone dust will settle a bit everywhere on plants and furnishings … No problem: the next day you will see nothing at all. Most of it will be eaten by snails!
Other sources of Calcium
Calcium for gastropods and for any other animal, must also be supplied with the food. Boiled vegetables such as zucchini and broccoli will be an excellent substitutes for cuttlefish bone.
Another way, as we have explained not as good as this, is the boiled egg shell.
Can powdered bone hurt fishes?
This is the most common question. When we dip the bone in the aquarium, the fishes may taste it and eat a lot of bone powder. How can we avoid this?
Simple: powdering the bone with lights off! During the night fishes sleep, while the snails go around eating all the bone available!
Does the cuttlefish bone modify the hardness in the aquarium?
One gram of bone completely dissolved in water gives on average 1.5 grams of Bicarbonate (the Carbonate becomes Bicarbonate, increasing by volume and weight) that, in a 100 liter aquarium, corresponds to about 15 mg/L of Calcium Bicarbonate.
Calcium Bicarbonate is composed of 75% Bicarbonate and 25% Calcium. Since 1 dKH corresponds to about 22 mg/L of Bicarbonate, for every 2 grams of bone we will have an increase of about 1 dKH.
In the same way, if 1 dGH corresponds to about 7 mg/L of Calcium, for every 2 grams of bone we will have an increase of about 1 dGH.
It will also increase around 80 μS/cm the conductivity of the water.
Bone and sparkling water
Another method to provide Calcium is to dissolve a couple of cuttlefish bones in a bottle of sparkling water. Why with gas? Because CO2 makes the water acid that helps to dissolve the cuttlefish bone.
We will put the grated bone in the bottle of sparkling water. If it dissolves immediately, we will put some more. Then we will shake and leave it in the fridge for 6 hours or in a freezer for half an hour.
Maybe you are wondering why we should keep our mixture in a refrigerator or freezer? Because the colder the water, the richer in CO2 it is!
Let’s look at the following image:
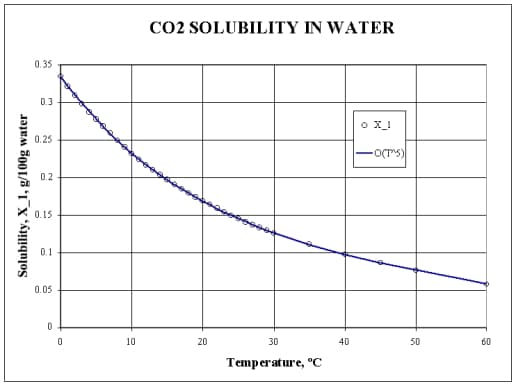
Water at refrigerator temperature (5 °C) contains almost twice as much CO2 as at the room temperature (20 °C). So it’s more acid, and it melts the cuttlefish bone better.
After these few and simple operations, our “raising hardness” drink will be ready to be poured into the aquarium.
Curiosity
Known in China under the name of Hai Piao Xiao, the cuttlefish bone is used in traditional Chinese medicine to treat gastritis, as well as for the treatment of duodenal ulcer and asthma. Surgeons consider it an excellent means of limiting bleeding in case of dental removal or in case of nasal septal surgery. And in case someone has … flatulence … cuttlefish bone! And the world will thank you! At least it is what the Chinese doctors say …
It is also appreciated by jewelers as it is a great mold, easy to carved and very robust thanks to its natural heat resistance.


In western pharmacology, Calcium Carbonate is used to neutralize stomach acidity, change the pH of gastric contents and promote ulcer healing.
In aquarium the bone can be safely used to saturate Akadama. We know that this type of substratum absorbs carbonates and then lowers the pH of the water. Increasing acidity dissolves our bone by releasing other carbonates. These will be absorbed by Akadama and so on until the saturation is complete.
I conclude by thanking all the users who contributed to the drafting of this article and inviting you to subscribe to our forum Acquariofilia Facile for any comment or clarification about it.
I remember the registration is easy and extremely fast!